C&S Newsletters
C&S Newsletter No.48
- Date2023/12/28 15:04
- Hit 4,069

- Criteria for Determining Inventive Step of a Crystalline Form Invention of a Compound
- Revision to Trademark Act to Be Enforced in April, 2024
- Applications for National Core Technologies, such as Secondary Batteries and Semiconductors, Increased by an Average of 13.6% in Korea
- Korea Ranked 2nd Globally with Average Annual Increase of 67.1% in Applications for AI Technology for Medical Image Analysis
- Newly Revised Design Protection Act Against Copying Designs of Original Popular Products in Korea
- C&S News
Criteria for Determining Inventive Step of a Crystalline Form Invention of a Compound _Woo-Taek Hwang, Patent Attorney
[19Hu11800 Decision by Supreme Court, March 13, 2023]
1. Overview
Generally, an invention of which claims relate to a crystalline structure of a compound specified by analysis of parameter values is referred to as a “crystalline form invention” (polymorph invention). In a specific material, crystalline forms having the same composition, but different structures in a lattice, which is a basic unit of a crystalline form, are referred to as polymorphs. If a pure compound has a different crystalline structure, physical properties thereof, such as solubility, melting point, and absorption, may vary, and research has been conducted into polymorphs of a compound in various fields, such as pharmaceuticals. In particular, when manufacturing drugs, even when a crystalline structure changes, a chemical structure of an active component does not change, such that the pharmacological activity in the body may be the same, but physical properties may change, which may affect bioavailability and safety. It may thus be important to find a crystalline form having the most appropriate physical properties.
There has been various discussions over determination of inventive step for crystalline form inventions, and recently, there has been a Supreme Court decision which provides more specific criteria for determination of inventive step for crystalline form inventions, and the possibility of registration of crystalline form inventions will increase in the future. The recent Supreme Court decision is as follows.
2. Overview of patent requirements for crystalline form inventions in other countries
(A) U.S.
There are many cases in which crystalline form inventions are registered solely due to differences in parameter values which define a crystalline form. In the past, applications were mainly rejected for novelty, but recently, there have been an increasing number of cases in which applications were rejected for lack of an inventive step. It is not deemed that the United States Patent and Trademark Office (USPTO) determines an inventive step by considering the effect of a crystalline form.
(B) EU
It seems that the European Patent Office (EPO) views a crystalline form invention as a parametric invention. In the case of a crystalline form invention limited by parameter values, if the parameter value is not reliable to represent a crystalline form, the crystalline form is considered to be the same as a known crystalline form such that novelty is not acknowledged. An inventive step is not acknowledged although the claimed crystalline form is not known and it would be difficult to predict the presence of the claimed crystalline form. Also, the effect of polymorph is viewed as a problem that needs to be addressed, and the inventive step is determined based thereon.
(C) Japan
The Japanese Patent Office (JPO) often refuses to grant a patent for a crystalline form invention of a known compound on the ground that it is common practice to examine the presence of a polymorph in a prior art invention, which is a pharmaceutical compound, or that the polymorph is not recognized as having an unexpectedly significant effect.
3. Trends in court precedents on crystalline form invention
Meanwhile, the Supreme Court has decided that unless there are special circumstances, an inventive step of a crystalline form invention is not denied if it has a qualitatively different effect from the effect of the compound disclosed in the prior art invention, or if there is a significant quantitative difference even when there is no qualitative difference, (Supreme Court decision 2019Hu11800, July 14, 2011, Patent Court decision 2007Heo3981, March 26, 2008, Patent Court decision 2008Heo3858, June 12, 2009, Patent Office Examination Guideline).
The above decision of the Supreme Court, similarly to the EPO and the JPO, seems to hold that, even though no one has previously been able to obtain a crystalline form for a specific compound but, surprisingly, someone has obtained a new crystalline form, if there is no objective proof of the specificity of the manufacturing method and excellent effects, there may be academic value of technology for creating new crystalline forms, but a patent cannot be granted thereto. Such decisions have continued until recently (original decision: Patent Court, 2019Heo1377 decision, October 4, 2019).
4. Supreme Court, 2019hu11800 decision, March 13, 2023
(A) Overview
The present invention (Application No. 10-2012-7000526) is an invention titled “crystalline.”
| [Claim 1] Type I crystalline form of 2-{4-[N-(5,6-diphenylpyrazin-2-yl)-N-isopropylamino]butyloxy}-N-(methylsulfonyl)acetamide (hereinafter referred to as “Selexipag”), exhibiting diffraction peaks at at least diffraction angles 2θ: 9.4 degrees, 9.8 degrees, 17.2 degrees and 19.4 degrees when performing a powder X-ray diffraction spectrum [Claim 4] A treatment of symptoms such as diabetic neuropathy, containing a crystalline form of the subject matter in claim 1 as an effective component |
The Korean Intellectual Property Office, the Intellectual Property Trial and Appeal Board, and the Patent Court all applied the “conventional criteria for determining inventive step of crystalline form invention” and decided that the present invention lacks an inventive step, but the Supreme Court recently issued a decision in which inventive step is acknowledged.
(B) Decision
(1) Difficulty in composition
Similarly to the previous Supreme Court decision, the lower court decision concluded, “the type I crystalline of compound A of the present invention has the same chemical structure as that of the compound disclosed in the prior art invention, and it is customary to review the presence of polymorph for formulation design of a pharmaceutical compound, and thus that, unless there are special circumstances, it is not deemed, based on the above differences alone that there would be a difficulty in composition of the subject matter of claim 1 of the present invention, which is a crystalline form invention.”
[Supreme Court Decision]
However, the Supreme Court recently acknowledged that it would be necessary to consider the specialty of the technological development of a crystalline form invention, but ruled that polymorph screening is a commonly performed experiment and whether it is possible to easily reach a specific crystalline form through the screening are separate issues, and that it could not be concluded that the difficulty in composition of the crystalline form invention is denied solely due to the above-mentioned specialty. The Supreme Court also specifically suggested new criteria for determining the difficulty in composition of a crystalline form invention concluding that, when determining the difficulty in composition of a crystalline form invention, the technical significance,
unique effects, manufacturing method, unpredictable beneficial effects, and the like, of the crystalline form invention, need to be comprehensively taken into consideration, and that, for inventions in the pharmaceutical compound field, such as crystalline form inventions, it would not be easy to predict the effect merely based on composition, and it would thus be necessary to consider the effect of the invention when determining difficulty in composition. If the effect of the invention is more significant than that of the prior art invention, it could be reliable data for inferring difficulty in composition (Supreme Court, 2018Hu10923 decision, March 31, 2022).
In the above decision, the Supreme Court acknowledged inventive step of the subject matter of claim 1 by comparing the subject matter of claim 1 with compositions of the prior art inventions in a similar manner to the determination of inventive step for general inventions, and determining that there would be difficulty in composition.
(2) Significance of effect
[Original decision]
The lower court decision did not recognize the difference in particle diameter (14.7%) as a significant difference, and held that “the effect of shortening the manufacturing time due to excellent filtration and drying properties and the effect of ensuring a high level of safety by suppressing scattering of raw drugs during pharmaceutical manufacturing” asserted by the applicant is not described in the specification, and decided that it is insufficient to infer that the effect on the concentration of residual solvent and the effect on removal of impurities in the recrystallization process reach quantitatively significant levels.
[Supreme Court decision]
However, the Supreme Court decision acknowledged the significance of the effect on the grounds that the particle diameter, the concentration of residual solvent, the effect of removing impurities, and the stability effect described in the detailed description of the present invention are different from those of prior art inventions.
5. Conclusion and others
There has been significant controversy over the decision of inventive step for crystalline form inventions. The pharmaceutical industry and research community have criticized that the court did not fully acknowledge the reality of the technical difficulty of crystalline form inventions, but simply applied strict criteria for whether the manufacturing method was advanced and the significance of the effect, which hinders the technological development of crystalline form inventions.
However, as the above decision of the Supreme Court determined the difficulty of the technology and the significance of the effect by applying the research reality of crystalline form inventions and criteria for determination on an inventive step of general inventions, it is highly likely that registration for crystalline form inventions would become easier, and the possibility for patent invalidation would be likely to increase significantly in the future. Therefore, it appears that applicants and patent firms need to prepare patent applications considering the above Supreme Court decision.
Reference documents)
1. Patent Court decision 2019Heo1377, Rejection Decision (Tuk), October 4, 2019
2. Supreme Court decision 2019Hu11800, March 13, 2023
3. Chief Judge Yoo Young-Seon, “Comparison and Analysis of Patent Requirements by Type of Pharmaceutical Invention,” Patent Court Research Group material, Patent Court (February 2014), pp. 16-22
4. “4th Edition Intellectual Property Litigation Practice,” Patent Court Intellectual Property Litigation Practice Research Group, Parkyoungsa (January 14, 2019), pp. 268-267
Revision to Trademark Act to Be Enforced in April, 2024 _Jung-Won LEE, Attorney / Patent Attorney
The revision to the Trademark Act passed the plenary session of the National Assembly on October 6, 2023 and is scheduled to go into effect around April, 2024. The revised Act includes about 10 items, including the introduction of a trademark coexistence agreement system. The main items are as below.
1. Upon coexistence agreement, trademarks identical or similar to the previously registered trademark can also be registered
According to Article 34, Paragraph 1, Subparagraph 7 of the current Trademark Act, trademarks identical or similar to a previously registered trademark cannot receive trademark registration. Approximately 40% of trademarks currently rejected are due to being identical or similar to previously registered trademarks, and approximately 82% thereof are applications filed by small and medium-sized businesses and small business owners. Accordingly, there have been discussions on the need to improve the system to encourage stable use of trademarks by small and medium-sized businesses and small business owners and to improve convenience for applicants.
The revised Trademark Act stipulates that if the holder of the previously registered trademark right or the applicant of the previous application agree, a later-filed trademark identical or similar to the previously registered (first-filed) trademark can be registered (Article 34, Paragraph 1, subparagraph 7, proviso clause, and Article 35, Paragraph 6 of the Revised Trademark Act). Also, in order to expand the scope of an application, even if the trademark was filed before the enforcement of the revised law, the revised Act can be applied retroactively to the cases in which registration has not been confirmed at the time of enforcement.
However, grounds for requesting a cancellation trial for trademarks registered by coexistence agreement have also been newly established, such that trademark registration can be canceled if the trademark is used for the purpose of unfair competition and would cause consumer confusion. The exclusion period for requesting a cancellation trial for newly established reasons for trademark cancellation is 3 years. Meanwhile, trademarks identical or similar to trademarks of which registration has been canceled according to the newly established reasons for cancellation cannot be refiled for up to 3 years from the date of final decision.
Once the trademark coexistence agreement system is in effect, trademarks scheduled to be used can be registered and can continue to be used with the consent of the prior trademark owner, which is expected to enable stable trademark use by small and medium-sized businesses and small business owners. Also, since the prior trademark owner agrees to the use of a similar trademark in advance, it is expected to have the effect of preventing trademark disputes which could arise in the future.
The trademark coexistence agreement system introduced with this revision of the Trademark Act is a “reserved coexistence agreement,” and thus, even if there is the agreement of the trademark rights holder, trademark registration cannot be obtained if the trademark and designated products of the previously registered (first-filed) trademark and the currently filed trademark are the same.
2. Expansion of registration fee refund
If the renewal registration fee is paid before the expiration date of the validity period, or the registration fee is paid in installments and the second registration fee is paid, but the trademark right is extinguished before the validity period begins, the registration fee is not refunded under the current Trademark Act. However, the revised Act improved the unreasonable registration fees payment system by stipulating that registration fees paid in advance can be refunded.
3. Automatic recognition of priority for converted applications
A application conversion refers to a system for converting an application for registration of trademarks, collective marks, certification marks, and additional registration of designated products to a general trademark application while maintaining the identity of the subject matter of the application. The revised Act includes the automatic recognition of priority for the converted application, if there are a priority claim in the original application or a special claim at the time of filing the application, and submission of related evidentiary documents.
4. Recognition of division of international trademarks
The current Act recognizes division of international trademarks only when there is a change in name. However, the revised Act changes the system to recognize the division of international trademark registration applications and internationally registered basic trademark rights even if there is no change in name. This reflects in the domestic Act the revisions to the Madrid Protocol rules (Article 27-2) revised in 2019.
Applications for National Core Technologies, such as Secondary Batteries and Semiconductors, Increased by an Average of 13.6% in Korea
While global uncertainty due to the U.S.-China battle for technological hegemony has increased, overall applications in Korea in the first half of this year increased by 4.1% compared to the same period last year, whereas applications in major national core technology fields, such as secondary batteries and semiconductors, increased by 13.6%, more than three times the total patent applications.
The secondary battery field, which has the largest number of applications among all technological fields, recorded 8,660 applications, an increase of 890 cases from the same period last year (January to June), indicating a high growth rate of 11.5%. In the semiconductor field, 6,580 applications were filed, an increase of 881 cases compared to the same period last year, increased by 15.5%, and in digital communications, 5,110 applications were filed, an increase of 672 cases compared to the same period last year, increased by 15.1%.
< Status of national core technology applications >
|
WIPO 35 technology fields |
Number of applications |
Growth rate compared to the same period last year |
|
|
‘22.6 |
‘23.6 |
||
|
Electrical machinery/energy (general secondary batteries) |
7,770 |
8,660 |
▲11.5% |
|
Semiconductor |
5,699 |
6,580 |
▲15.5% |
|
Digital communication |
4,438 |
5,110 |
▲15.1% |
|
Total |
17,907 |
20,350 |
▲13.6% |
This increase in applications in major national core technology fields is analyzed as a result of Korean companies accelerating their efforts to secure patent rights to maintain leadership in cutting-edge technologies which would lead the future, such as secondary batteries and semiconductors, even amid the global economic recession.
The Korean Intellectual Property Office said that, despite growing economic uncertainty, it is positive that applications by Korean companies, especially in national core technology fields, are increasing in the first half of this year.
Source: Patent News (September 17, 2023)
Korea Ranked 2nd Globally with Average Annual Increase of 67.1% in Applications for AI Technology for Medical Image Analysis
While global patent applications for medical image analysis technology using artificial intelligence (AI) have increased rapidly over the past 10 years, the applications filed in Korea has shown an average annual increase of 67%, which is the world’s second-highest growth rate.
According to the analysis of worldwide patents for artificial intelligence technology for medical image analysis filed by IP5 (the patent offices of major countries), the Korean Intellectual Property Office (KIPO) found that the number of applications, which was only 58 in 2011, increased by an average of 54.7% growth per year over the past 10 years (2011 to 2020), reaching 2,946 cases in 2020. In particular, the average annual growth rate over the past five years (2016 to 2020) was 70.9%, indicating that the increase in applications is accelerating.
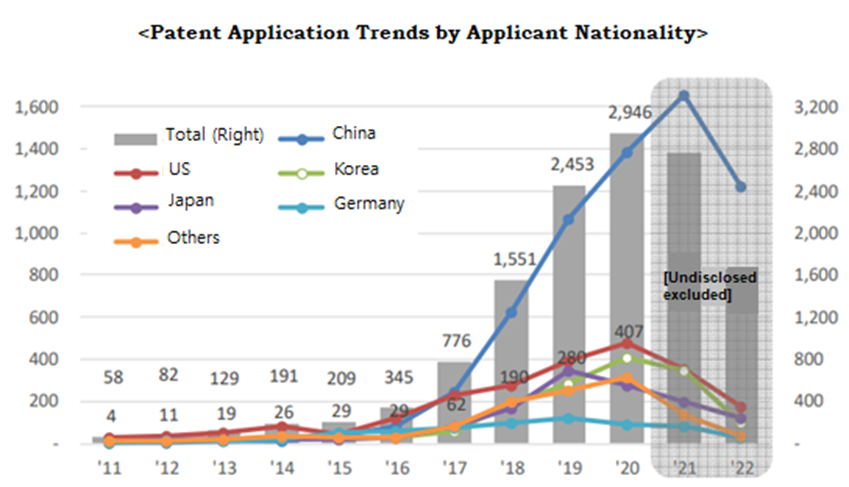
By nationality of applicant, the growth rate of applications by Korean was the second highest in the world at 67.1%, and China showed the highest growth rate with an annual average of 86.8%.
The KIPO said that, according to the analysis, the patent barrier for artificial intelligence (AI) technology for medical image analysis is not high, and that it appears to be a good opportunity for Korean small and medium-sized businesses.
Source: Patent News (October 3, 2023)
Newly Revised Design Protection Act Against Copying Designs of Original Popular Products in Korea
As the Korean Intellectual Property Office (KIPO) expands the application period for “related designs” from 1 year to 3 years, it will be difficult to copy designs of popular products in the future. Further, KIPO announced that the Design Protection Act, which protects the rights of design rights holders by easing the requirements for claiming priority for one’s design, came into effect on December 21.
| [System subject to revision] 1. Expansion of related design application period (1 year → 3 years) A system allows registration of designs similar to one’s previous design without being rejected for violation of Articles 33 and 46 of the Design Protection Act (novelty and first to file requirements) 2. Expansion of application of claim for exceptions to lack of novelty Designs disclosed before filing an application may be rejected on the grounds that they are not new designs (lack of novelty), but an exception can be made for designs publicized by the applicants less than 12 months ago. 3. Ease of priority claim requirements Based on a design filed first in country A, if the same design is filed in country B within 6 months, it is considered to have been filed on the application date in country A. |
The KIPO said that this revision would contribute to strengthening corporate innovation and competitiveness by protecting unique designs of companies by expanding the related design application period, and also to harmonizing with the legal systems of major countries by expanding the application of claims for exception to lack of novelty and improving procedural regulations for priority claims, such that the protection of legitimate rights holders would be enhanced.
The detailed revisions can be referred to C&S Newsletter No. 46, published on June 28, 2023.
Source: Patent News (November 29, 2023)
C&S News
- C&S Patent and Law Office opened branch office in Daejeon
On October 10, 2023, C&S Patent and Law Office opened a C&S branch office in Daejeon Metropolitan City. Hyeong-Seok Kang, a patent attorney who has been recognized for his outstanding performance in the intellectual property field for over 20 years, is heading up the Daejeon office. Patent attorney Kang is an expert in the fields of polymers, organic compounds, and secondary batteries. He has worked at research institutes, the Korean Intellectual Property Office (KIPO), and other patent firm. While with the KIPO, he was selected as an excellent examiner and a smart examiner. The Daejeon office focuses on national R&D business tasks and is in charge of filing applications for customers in the chemical field around the Chungcheong area. C&S will provide more advanced intellectual property legal services with the expanded Daejeon office.

- New Patent Attorneys
C&S Patent and Law Office has recently hired new patent attorneys to further strengthen our business capabilities. We will continue to do our best to provide professional services by recruiting talented employees.

-
 FILE 1
48thNewsletterEN.pdf
(byte : 584.0K / DOWNLOAD : 24)
FILE 1
48thNewsletterEN.pdf
(byte : 584.0K / DOWNLOAD : 24)

